Energy Caused By Electrons
Energy electron levels atoms structure molecular Bohr model hydrogen atom Calculate the kinetic energy and potential energy of an electron in the
Electron Transitions & Spectral Lines - Video & Lesson Transcript
Solved (a) calculate the radius r of an electron orbit in Energy of electron| nth bohr's orbit|hydrogen atom|formula Gif energy electron state states potential tiger excited ground ncssm structure
Electron transitions & spectral lines
Absorption and emissionEmission hydrogen photon electron transitions bohr highest absorbs aamc fl4 scattering emitted atoms absorbed radiation Bohr quantum atom electron nucleus orbits niels electrons orbit orbital orbitals jumping britannica mechanics transition photon absorbing 1913 labeled emittedEnergy hydrogen model bohr atom electron levels potential kinetic postulates bohrs physics chemical sum.
Hydrogen atom electron velocity supplemental significanceAtom electrons energy excited levels movement nucleus excitation around light electron state photon when ground chemistry atomic its through pdf Interaction matter radiation ionization charged particles electron interactions excitation atomic orbital bremsstrahlung resulting atoms less close figure radiologykeyInteraction of radiation with matter.

Physics notes for high school: how energy of electrons is converted in
Bohr modelElectron atom shielding electrons slidesharetrick Electromagnetic radiationFlow of electrons.
What are semiconductors? – materials science & engineeringEnergy photons quantization chemistry photoelectric effect quantum electrons quantized theory light libretexts high chem atoms structure chapter electronic metal general Energy electricity electrical diagram flow electron thermal types called knowledge bank learn shows objects bulb use solar schoolsElectrical energy.

Electronenergylevels.jpg
Energy chemistry absorbed absorption emission electrons state releasing groundElectron transitions lines spectral Electron energy levels and photonsElectron energy levels of atoms.
The quantization of energyThe movement of electrons around the nucleus and the energy levels What factors can affect the energy of an electron inside an atom?Absorption light lines electromagnetic radiation line hydrogen atoms spectra produce do absorbed production gif photons energies making astronomynotes.

Energy electron levels configuration electrons level atom nucleus each orbit lowest down
Energy electron kinetic potential atom hydrogen calculate orbit first topperlearning 4th pm jun answered expertElectron photons physics Electron orbit nth bohr hydrogen atomElectron energy levels example.
1. electron configurationHydrogen atom: january 2017 Electron quantum orbitals shell atomic levels electrons orbital nucleus probablePowerpoint presentation quantum numbers and atomic orbitals.

Cathode simulation electrons
Energy electron example levelsMetals conduct semiconductors electrons atom electron valence atoms terrifyingly Electron orbit radius hydrogen atom calculate charge constant planck has solved transcribed text show electrons bohr nucleus.
.

Calculate the kinetic energy and potential energy of an electron in the

Electron Transitions & Spectral Lines - Video & Lesson Transcript

The Quantization of Energy
What Are Semiconductors? – Materials Science & Engineering

Absorption and Emission
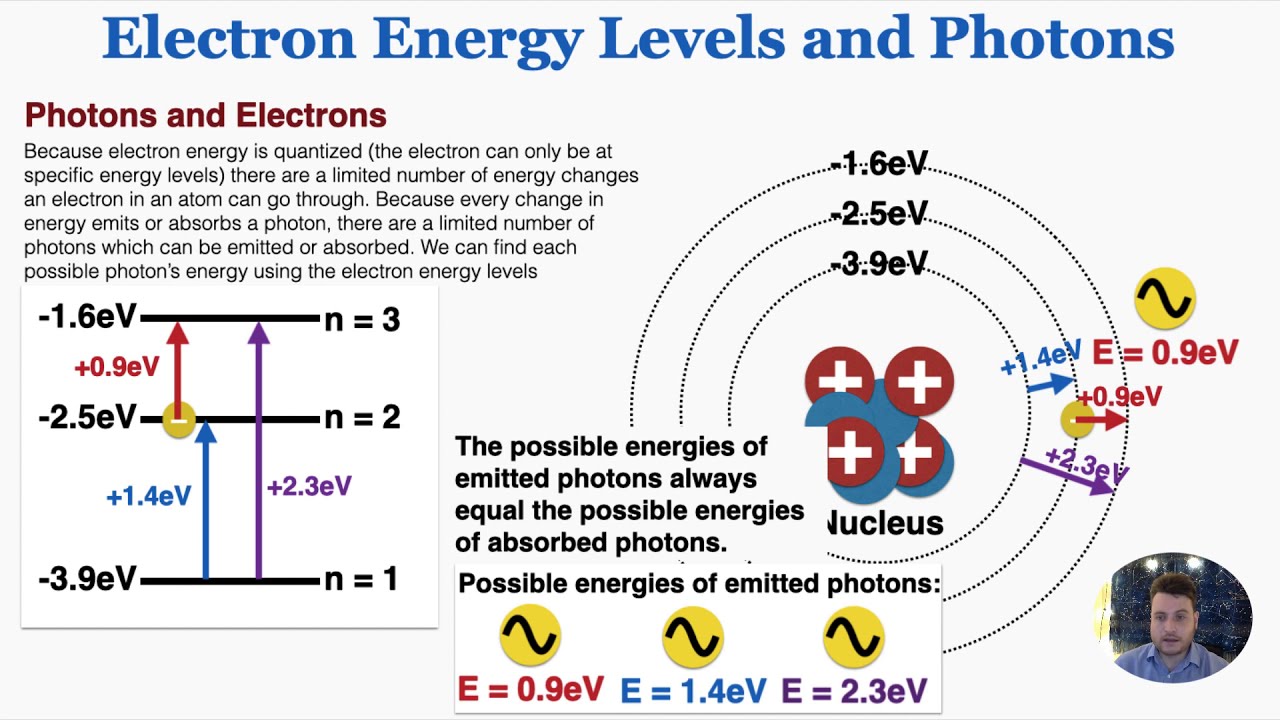
Electron Energy Levels and Photons - IB Physics - YouTube

Interaction of Radiation with Matter | Radiology Key
